
When immunized, these animals generate antigen-specific, fully human monoclonal antibodies. The mice are produced by introducing gene segments of human immunoglobulin loci into the germlines of mice deficient in mouse antibody production due to targeted gene deletions. The creation of transgenic mice strains engineered to produce human antibodies instead of mouse antibodies was a breakthrough in the therapeutic monoclonal antibody development. They are recognized by the patient’s immune system as foreign proteins, stimulating the production of human anti-mouse antibodies (the HAMA response) and resulting in a short serum half-life, which may limit their therapeutic use and cause possible allergic reactions. Murine mAbs produced from hybridomas can have high specificity, but do not always trigger human effector functions (e.g., complement systems). The mouse or rat part of the antibody binds the antigen while the humanized part helps the molecule evade the body’s immune system. A humanized monoclonal antibody consists of a human antibody combined with a small part of a murine monoclonal antibody. When monoclonal antibody development is intended for downstream therapeutic purposes, the best course of action is often to create a humanized monoclonal antibody. There are approximately 100 therapeutic mAbs on the market in the US, with many more in development and trials. Monoclonal antibody development therapeutics have been rapidly increasing their market share over the last decade, and in 2019, six out of the ten top-selling drugs were mAbs. This has the advantage of minimizing adverse side effects, especially when highly toxic drug substances must be delivered. For example, therapeutic mAbs exhibit much higher specificity than small molecule drugs. 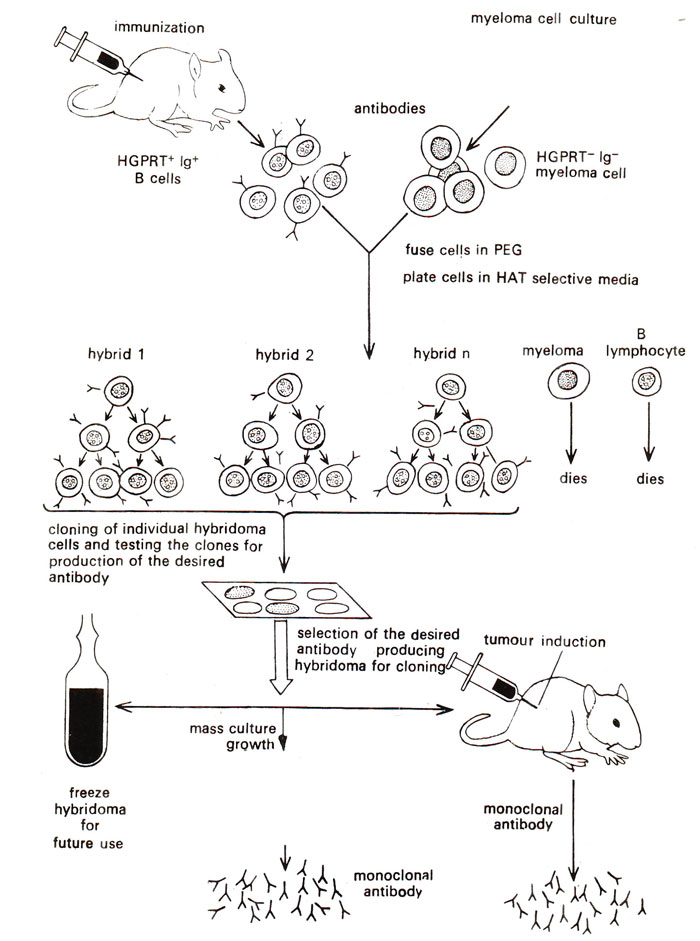
Monoclonal antibody development provides a number of advantages for therapeutic purposes. Newer technologies use molecular biology techniques to amplify antibody genes by PCR and recombinant technology for production in either bacterial or mammalian expression systems. Since the hybridoma technique was introduced by Köhler and Milstein in 1975, monoclonal antibody generation technologies have developed to include methods such as the use of transgenic animals to generate fully human mAbs antibody phage display and single B cell isolation followed by clonal production. Monoclonal antibody development occurs when hybridomas are propagated in cell culture to produce large quantities of antibody molecules that all bind to the same antigen. The resulting hybridoma cell line has the longevity and reproductivity of the myeloma and the antibody-producing ability of the B cell. These B cells are then fused with a myeloma cell (immortal B cell cancer cell) from a cell line that has lost the ability to produce immunoglobulins, but retains the ability to multiply indefinitely. The next step is to harvest the B cells that produce antibodies binding to the antigen. Hybridoma production starts with injecting a mouse with an antigen that provokes an immune response. Monoclonal antibodies are developed in bulk by a clonal population of hybridoma cells. This article introduces key topics in therapeutic mAb development. There are many factors to think about from the basic research behind selecting a target antigen to the various antibody generation methods available, characterization strategies, and commercial production. It can be a long and costly journey to find an effective therapeutic a study carried out on 63 drugs and biologic agents approved by the US Food and Drug Administration between 20 found that the estimated median capitalized research and development cost per product was $985 million. In 2020, there are approximately 100 therapeutic mAbs on the market in the US, with many more in development and trials. Humira therapy, a rheumatoid arthritis drug, sales topped $19 billion, while Keytruda therapy, a cancer drug which forms part of multiple immunotherapy strategies, had sales of >$11 billion. Monoclonal antibody therapeutics have been rapidly increasing their market share over the last decade, and in 2019, six out of the ten top-selling drugs were mAbs. Therapeutic mAbs exhibit much higher specificity than small molecule drugs, which minimizes adverse side effects, especially when highly toxic drug substances must be delivered. These differ from polyclonal antibodies (pAbs), which are heterogeneous mix that recognize and bind to multiple epitopes of a single antigen.They are employed in immunotherapies in one of several ways: Stimulate the patient’s immune system, Modulate or inhibit a biochemical disease pathway to deliver a therapeutic radionuclide or drug to a target cell type. Monoclonal antibodies (mAbs) are antibodies produced by a clonal population of B cells and bind to a single target antigen.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
